Can Polymers Be Both Tough and Trashy? Osaka Scientists Say Yes!
Polymers are everywhere—from your phone case to plastic water bottles. But here’s the catch: they need to be sturdy to do their job, yet easy to break down when tossed out. Yet we also want polymer degradation that vanish later. Usually, they are either tough or biodegradable—doing both well was thought impossible. Now, some brainy researchers at The University of Osaka cracked that problem wide open! A Light Switch Makes It Disappear? So, if you’re a high school student curious about cool science that actually changes the world, this story is just for you.
Key Takeaways
- Light can switch polymer degradation on or off.
- Movable cross-links slide for toughness and flexibility.
- Enzymes finish the job after light exposure.
- Spatial control allows patterns like a QR code.
- This could reshape medical and smart material design.
This material blends three big ideas. It uses light response, movable rings, and enzymatic segments. To explain, these parts work together. As a result, the plastic lasts, then degrades safely.
What did the Team Actually Build?
- A polyester network with movable cross-links.
- Cross-links use cyclodextrin rings that slide.
- Sliding reduces stress and improves toughness.
- Light changes host–guest interactions in the network.
- Enzymes access segments only after light exposure.
At this instant, think of rings as tiny sliders. With attention to stress, they move along chains. So that, the network avoids sudden breaks. To put it differently, the material bends and resists failure.
Contents
ToggleThe Science of Polymer Degradation in Plain Words
- Cyclodextrin rings are sugar-based molecules.
- They thread and slide on polymer chains.
- Light repositions rings, exposing sensitive spots.
- Enzymes, like lipases, then cut polyesters.
- Non-irradiated areas stay stable and strong.
By and large, that is the magic. Light gives precise control here. In this case, you choose what should degrade. To sum up, the rest stays strong and useful.
Why Should Students Care abut Polymer Degradation?
- This solves a major trade-off in materials.
- It links physics and chemistry you study now.
- It shows real lab methods and applications.
- It opens exciting careers in materials science.
- It connects biology via enzymes to polymers.
At this point, you can map class ideas. With this intention, think bonds, networks, and catalysis. As an illustration, link light energy to reactions. In short, this is textbook science in action.
Where Might This Help?
- Smart implants with controlled lifetimes.
- Coatings that degrade on schedule.
- Information materials with hidden patterns.
- Recyclable parts with on-demand breakdown.
- Classroom demos of light-driven change.
To enumerate, picture a medical stent. At the present time, removal can be hard. With this in mind, light could time its dissolution. In effect, patient care becomes gentler and smarter.
How the Light Switch Works for Polymer Degradation
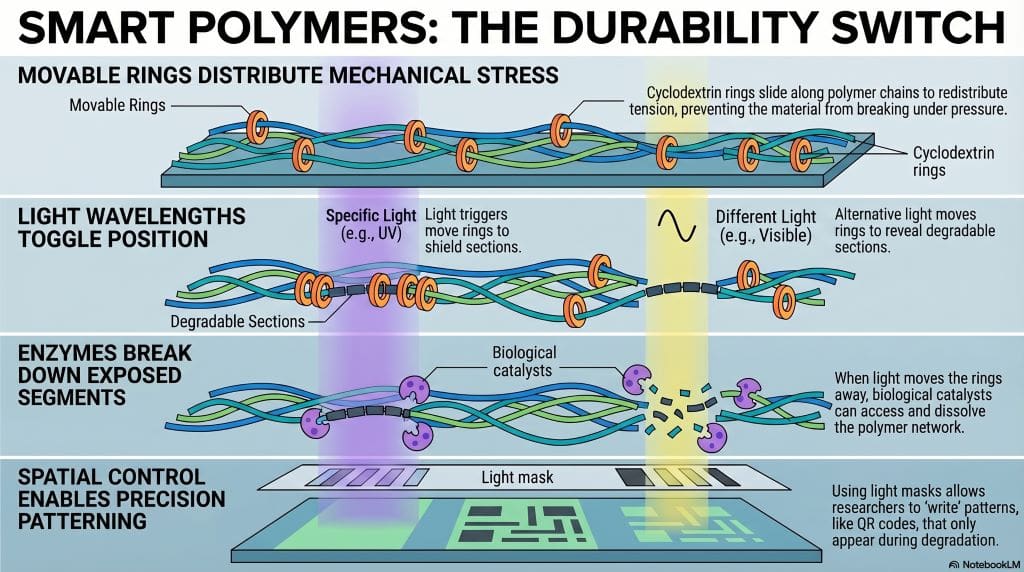
- Light shifts ring positions on chains.
- Rings either shield or expose segments.
- Exposed segments meet enzyme action.
- Shielded parts remain intact and tough.
- Spatial masks control patterns precisely.
All of a sudden, you can “write” a QR code. After that, enzymes read the lighted pattern. With the result that, the code appears by degradation. To rephrase it, light programs the material’s fate.
The QR Code Demo
- A photomask blocks light in places.
- Exposed areas get activated for enzymes.
- Degraded parts reveal clear visuals later.
- The pattern stays sharp and localized.
- It proves precise space-time control.
To the end that students see it, this is cool. In detail, it shows design and verification. To say nothing of clever thinking, it inspires. At last, research meets classroom curiosity.
Skills and Paths for You
- Learn polymer chemistry basics now.
- Practice spectroscopy and testing tools.
- Explore enzyme biochemistry connections.
- Build coding for data analysis.
- Join lab internships or camps.
With this purpose in mind, plan your courses. In like fashion, mix physics with chemistry. On one hand, take biology for enzymes. On the other, sharpen math for modeling.
Helpful Resources
- Start with Polymers on ENTECH Online.
- Read career guides in materials pathways.
- Follow university lab pages and news.
- Practice reading open-access articles.
- Enter science fairs with polymer projects.
As has been noted, we connect class to careers. With this in mind, see our guide: Careers in Materials Science. To put it another way, begin with Polymers Basics. In general, start small and grow skills.
Where this Research of Polymer Degradation Came from
- Osaka University’s team led the work.
- It appears on their ResOU portal.
- The senior author is Yoshinori Takashima.
- The lead author is Xin Zhou.
- The work connects to ACS Nano publication.
Why movable cross-links matter
- Fixed cross-links resist movement under stress.
- That can cause sudden failure points.
- Movable links redistribute forces smoothly.
- The network survives bigger deformations.
- Toughness improves without losing control.
Balanced against rigid networks, this is different. With this intention, the rings slide under load. As a result, chains avoid tearing. All things considered, durability gets smarter.
Lab concepts connected to class
- Host–guest chemistry uses molecular fits.
- Light interacts with molecules and bonds.
- Enzymes catalyze specific reactions.
- Networks form through cross-linking.
- Mechanical tests measure stress and strain.
To point out, your classes cover these. Prior to college, master core **. To illustrate, try small projects at home. By all means, ask teachers for support.
A simple mini-project idea
- Make a gelatin gel with cross-links.
- Change conditions to soften the gel.
- Test simple loads and deformations.
- Track changes with a ruler.
- Record results in a notebook.
Although this may be true, gels aren’t polymers here. In like manner, they show network behavior. To put it another way, this builds intuition. Summing up, learn by small experiments.
Potential real-world impact
- Consumer goods with programmed lifetimes.
- Medical devices with gentle removal.
- Electronics with serviceable parts.
- Packaging that degrades on schedule.
- Educational kits for classroom learning.
At any rate, regulation will matter here. With attention to safety, tests must verify behavior. In effect, standards will guide adoption. In conclusion, engineering follows careful evidence.
Career Spotlight: Materials Scientist
- Work on smart polymers and gels.
- Design tests for durability and safety.
- Collaborate across chemistry and biology.
- Communicate findings to engineers.
- Translate lab discoveries to products.
To repeat, these roles need teamwork. With this in mind, practice clear writing. In similar fashion, train in statistics. In due time, you can build real solutions.
Frequently Asked Questions, FAQs
Toughness and degradability combine with light control.
No, only where light activates access.
Sliding rings redistribute stress under load.
They act after light exposes segments.
Chemistry, physics, biology, and math.
Osaka University scientists, including Takashima and Zhou.
All in all, this work feels practical and inspiring. With this intention, use it to steer studies now. To this end, explore labs and courses early. In essence, build skills for future impact.
Reference
Osaka University. (2026, March 25). Breaking the durability–degradability trade-off in polymers. ResOU. https://resou.osaka-u.ac.jp/en/research/Breaking-the-durability2013degradability-trade-off-in-polymers
(Information summarized from the open-access ResOU article provided above.)
- Author
- Latest Posts
Charudatta Pathak was a student at St. Aloysius High School in Bhusawal, established in 1874, making it one of India’s oldest convent institutions. He earned his bachelor’s degree in mechanical engineering from SSVPS College of Engineering, Dhule in 1995, demonstrating exceptional academic achievement. Thereafter, he launched his career as a CNC programmer in a toolroom. He has utilized FANUC, Deckel, Maho, and Mikron controllers to operate 2-axis and 3-axis machines for the fabrication of jigs, fixtures, dies, and molds. He commenced his career as an academic in 1996, when he assumed the role of lecturer at SSVPS College of Engineering, Dhule. Additionally, he earned his post-graduate degree in CAD/CAM from L. D. College of Engineering, Ahmedabad, in 2000, and then acquired a Ph.D. from COEP in 2011 while concurrently engaged in teaching. Until 2023, he occupied many academic roles, including lecturer, assistant professor, professor, dean, principal, director, and vice chancellor at both public and private universities throughout India. From 2008 to 2010, he served as the project lead in the CAE department of a European multinational firm. During his 28-year professional tenure, he identified a need for dependable books targeted at students in grades 8 to 12, particularly for nascent career planning. He founded ENTECH Digital Magazine, a free monthly publication available on entechonline.com and magzter.com. Teens with a strong interest in Science, Technology, Engineering, and Mathematics (STEM) who aspire to pursue careers in these fields may find ENTECH Digital Magazine beneficial.
- Can Polymers Be Both Tough and Trashy? Osaka Scientists Say Yes!
- 'Spin Flip Emitter' in metal complexes can help solar cells leap beyond limits
- Uber and Lyft Accidents in Bergen County What To Know
- Why Most STEM Career Advice Fails—and How to Actually Find Your Dream Job!
- This Giant Space Cloud Hid a Massive Secret for 24 Years... Until Hubble Looked Again!