The Structure of Atom: A Foundation of Chemistry
Atoms are the fundamental building blocks of all matter. They form everything we see around us. The study of atomic structure helps us understand chemical reactions and molecular behavior. Scientists have spent centuries unraveling the mysteries of these tiny particles. As a result, we now have a comprehensive understanding of atomic composition.
The Atom’s Basic Components
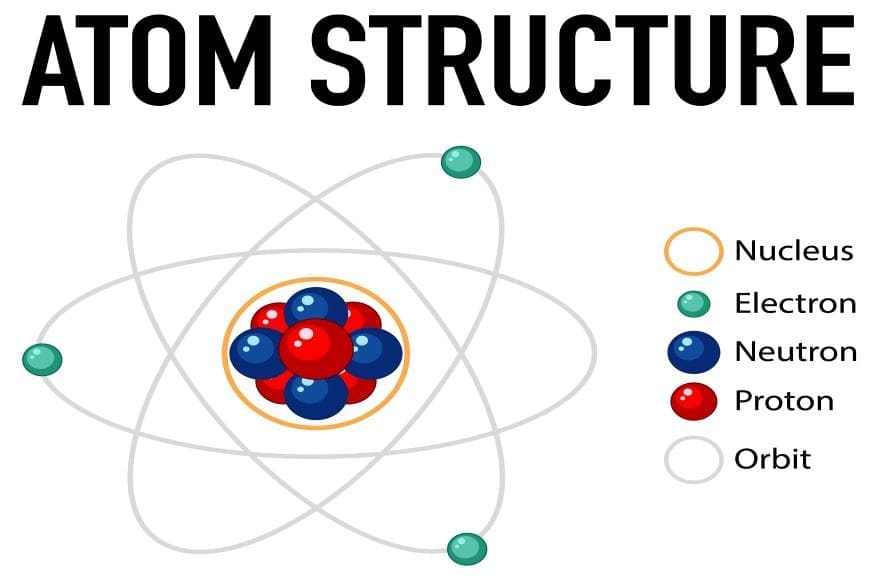
Every atom contains three primary subatomic particles. These particles are protons, neutrons, and electrons. Protons carry a positive charge. Neutrons have no electrical charge. Electrons possess a negative charge. All in all, these particles work together to form stable atoms.
Historical Development of Atomic Theory
Ancient Greek philosophers first proposed the concept of atoms. At this time, they believed matter could not be divided infinitely. John Dalton advanced this theory in the early 1800s. He suggested that atoms were solid, indivisible spheres. After that, J.J. Thomson discovered the electron in 1897. This discovery changed our understanding of atomic structure completely.
The Evolution of Modern Atomic Models
Ernest Rutherford performed his famous gold foil experiment in 1911. He discovered that atoms have a dense central nucleus. Niels Bohr later proposed that electrons orbit this nucleus. At the present time, we use quantum mechanical models for accuracy. These models describe electron behavior more precisely than earlier theories.
Also read: Most Important Functional Group
The Atomic Nucleus
Protons: The Identity Markers
Protons determine an element’s atomic number. The atomic number defines what element an atom represents. For example, all carbon atoms contain exactly six protons. Protons reside in the nucleus at the atom’s center. They contribute significantly to the atom’s total mass. What’s more, protons remain constant for each element regardless of isotopes.
Neutrons: The Stability Providers
Neutrons work alongside protons in the atomic nucleus. They add mass to the atom without affecting its charge. The number of neutrons can vary within the same element. That is to say, atoms with different neutron counts are called isotopes. Neutrons help stabilize the nucleus through the strong nuclear force. Without neutrons, many atoms would be unstable and radioactive.
Nuclear Forces and Binding Energy
The strong nuclear force holds the nucleus together firmly. This force overcomes the electrical repulsion between positively charged protons. Binding energy measures how tightly nuclear particles are held together. As can be seen, heavier elements require more binding energy for stability. The balance between protons and neutrons determines nuclear stability.
Electron Configuration and Arrangement
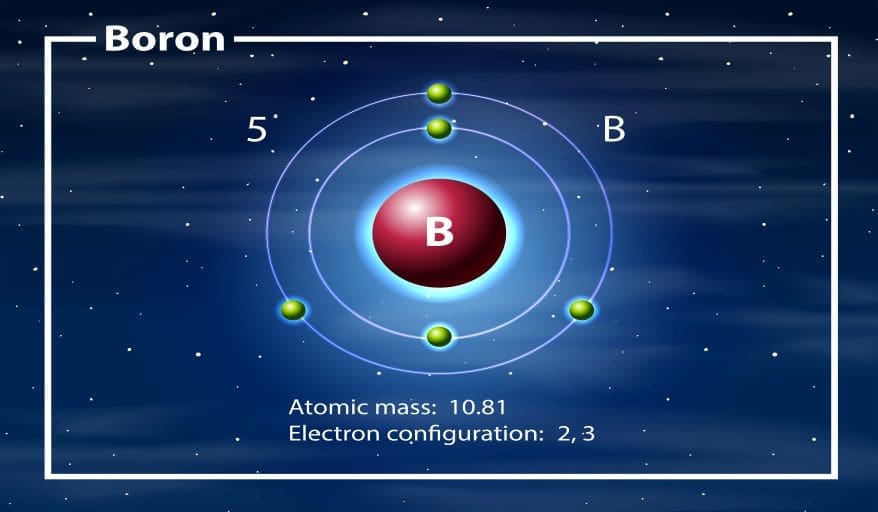
Energy Levels and Shells
Electrons occupy specific energy levels around the nucleus. These levels are often called shells or orbitals. The first shell can hold a maximum of two electrons. After that, subsequent shells hold more electrons according to specific rules. In other words, the maximum capacity follows the 2n² formula. Electrons fill lower energy levels before occupying higher ones.
Subshells and Orbital Types
Each energy level contains one or more subshells. The subshells are designated as s, p, d, and f. To illustrate, the first energy level has only one s subshell. The second level contains both s and p subshells. Moreover, each subshell type has a distinct shape and capacity. The s subshell is spherical and holds two electrons maximum.
The Aufbau Principle and Electron Filling
The Aufbau principle guides how electrons fill available orbitals. Electrons occupy the lowest available energy state first. This means they fill orbitals in a predictable order. At the same time, Hund’s rule states that electrons fill orbitals singly first. They only pair up after all orbitals in a subshell are half-filled.
Valence Electrons and Chemical Properties
Valence electrons occupy the outermost energy level of an atom. These electrons determine an element’s chemical reactivity and bonding behavior. To point out, elements in the same column share similar properties. This similarity occurs because they have the same number of valence electrons. Above all, valence electrons participate in chemical bond formation.
Quantum Numbers and Electron Description
Principal Quantum Number
The principal quantum number (n) indicates the electron’s energy level. It can be any positive integer value. As a matter of fact, higher values mean electrons are farther from the nucleus. These electrons also possess more energy than those closer in. The principal quantum number determines the shell an electron occupies.
Angular Momentum Quantum Number
The angular momentum quantum number (l) defines the orbital’s shape. It ranges from 0 to (n-1) for each energy level. For instance, when l equals 0, the orbital is s-type. When l equals 1, the orbital is p-type. This number also determines the number of angular nodes present.
Magnetic and Spin Quantum Numbers
The magnetic quantum number (m_l) specifies orbital orientation in space. It ranges from -l to +l in integer steps. In addition, the spin quantum number describes electron spin direction. Electrons can spin either up or down on their axes. Consequently, no two electrons can have identical quantum number sets.
Isotopes and Atomic Mass
Understanding Isotopic Variation
Isotopes are atoms of the same element with different neutron counts. They share identical proton numbers but differ in mass. To summarize, carbon-12 and carbon-14 are both carbon isotopes. Carbon-12 has six neutrons while carbon-14 has eight neutrons. Nevertheless, both contain six protons in their nuclei.
Atomic Mass Calculations
The atomic mass represents the weighted average of all isotopes. Scientists measure atomic mass in atomic mass units (amu). This measurement accounts for the natural abundance of each isotope. As an illustration, chlorine has two main isotopes in nature. The atomic mass reflects the proportional contribution of each isotope.
Conclusion
The structure of the atom forms the foundation of chemistry. Understanding atomic components helps explain chemical behavior and reactions. Scientists continue to refine our knowledge of atomic structure today. With this knowledge, we can predict and manipulate chemical processes effectively. The atom remains central to all chemical and physical phenomena.
Additionally, to stay updated with the latest developments in STEM research, visit ENTECH Online. Basically, this is our digital magazine for science, technology, engineering, and mathematics. Also, at ENTECH Online, you’ll find a wealth of information.
References
Salvi, P. R. (2025). On the Birth of Modern Chemistry: I—The Atomic–Molecular Theory from Dalton to Avogadro. Histories, 5(1), 8. https://doi.org/10.3390/histories5010008
Kragh, H. (n.d.). Before Bohr: Theories of atomic structure 1850-1913. RePoSS. https://www.ub.edu/hcub/hfq/sites/default/files/reposs-010.pdf