This Plastic-Boron Mix Blocks Deadly Neutrons — and It Could Change Nuclear Safety Forever
Think about the walls of a nuclear power plant. They must stop invisible particles. Fast neutrons are among the trickiest to stop. They zoom through most materials easily. Now, scientists Amina Hadjal and her team members have mixed everyday plastic with a special powder called titanium diboride. The result? A lightweight shield that blocks fast neutrons surprisingly well. This discovery of boron neutron shielding material connects directly to chemistry, physics, and materials science — subjects you study right now. And it could open exciting career doors for curious students like you.
Key Takeaways
- Researchers mixed polyethylene (PE) with titanium diboride (TiB₂) to make a neutron shield.
- The PE–30TiB₂ composite performed best, outperforming pure polyethylene significantly.
- The study used GEANT4 computer simulations to test neutron attenuation.
- Higher TiB₂ content meant more neutrons were blocked.
- This material is lightweight, making it practical for real nuclear facilities.
- The research connects directly to chemistry, physics, and materials science at Class 11–12 level.
What Are boron neutron shielding material and Why Should You Care?
At first, neutrons sound like a topic only for PhD scientists. But provided that you study atomic structure in Class 11 chemistry, you already know the basics. Neutrons are particles that live inside an atom’s nucleus. They carry no electric charge. That is actually what makes them so dangerous.
Why Neutrons Are Hard to Stop
To explain this simply — charged particles like alpha or beta particles interact with electric fields in matter. They slow down quickly. Neutrons, on the other hand, pass straight through most materials without being stopped. This is because they feel no electromagnetic force. Fast neutrons carry high kinetic energy and can damage living cells deeply.
- Alpha particles — stopped by a sheet of paper or skin. Very short range.
- Beta particles — stopped by a few millimetres of aluminium. Medium range.
- Gamma rays — stopped by thick lead or concrete. High penetration.
- Fast neutrons — extremely penetrating. Require specialized composite materials to stop them effectively.
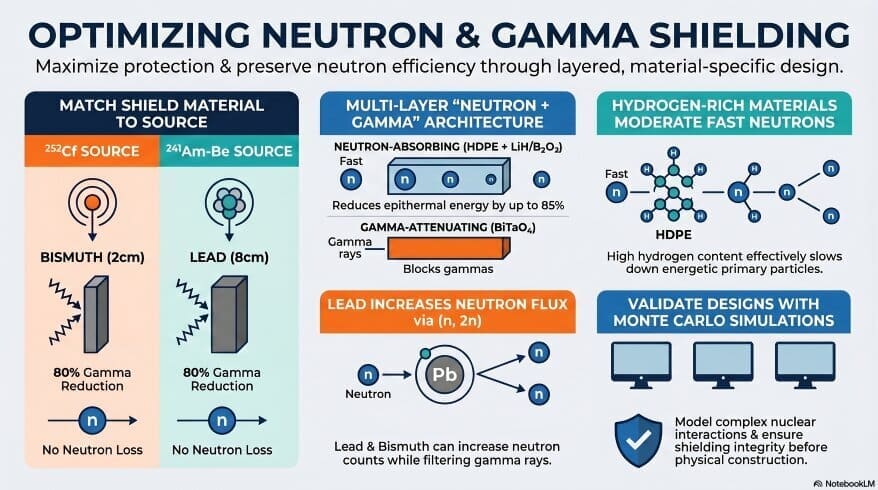
At the present time, nuclear facilities use concrete, water, and borated polyethylene as shields. Each has limitations, though. Concrete is heavy. Water needs containment. These challenges motivate scientists to develop smarter, lighter materials.
The Chemistry Behind the Discovery of boron neutron shielding material
So how does the new material (boron neutron shielding material) actually work? To put it differently, it is a two-step process happening inside one composite material. All things considered, this is where your Class 11 and 12 chemistry becomes directly useful.
Step 1: Polyethylene Slows the Neutrons Down
Polyethylene (PE) is the same plastic used in shopping bags and milk cartons. You study polymers and hydrocarbons in organic chemistry. As a matter of fact, PE is a long chain of carbon and hydrogen atoms. Hydrogen atoms are excellent at slowing fast neutrons. When a fast neutron collides with a hydrogen nucleus, it loses energy rapidly. This process is called moderation.
Think of it like this: A fast-moving billiard ball slows down most when it hits another ball of similar mass. Hydrogen nuclei (protons) are almost the same mass as neutrons. So collisions between them are very efficient at transferring energy — and slowing neutrons down.
Step 2: Boron Absorbs the Slowed Neutrons
After slowing down, the neutrons become “thermal” or slow neutrons. Here is where titanium diboride (TiB₂) does its job. Boron has an extremely high thermal neutron capture cross-section. In plain language — boron is very good at trapping slow neutrons and absorbing them. Your Class 11 periodic table chapter covers boron as a metalloid in Group 13. Seeing that you already know where boron sits on the periodic table, this discovery makes a lot more sense now.
- Polyethylene matrix — rich in hydrogen atoms, slows fast neutrons via repeated elastic collisions.
- Titanium diboride (TiB₂) filler — boron absorbs the now-slow thermal neutrons efficiently.
- Titanium contribution — titanium atoms scatter neutrons, adding another layer of attenuation.
- Combined effect — two mechanisms working together produce shielding better than either material alone.
What’s more, the researchers tested three versions of this composite. To enumerate, they used 10%, 20%, and 30% TiB₂ by weight mixed into polyethylene. The version with 30% TiB₂ performed best in all tests (Hadjal et al., 2026).
You can read more about the chemistry of radioactive elements and their real-world importance in this detailed guide on Nuclear Chemistry at ENTECH Online.
How Scientists Tested It — Without a Real Reactor
Here is something fascinating. The researchers did not build an actual nuclear shield and test it in a reactor. While it may be true that lab experiments are important, computer simulations are equally powerful today. They used a sophisticated toolkit called GEANT4 to simulate neutron behaviour mathematically.
What Is a Monte Carlo Simulation?
To illustrate — imagine throwing a million virtual dice to predict how neutrons would behave inside different materials. Monte Carlo simulations do exactly that. They run thousands of random neutron paths through a virtual material and calculate how many get through. This method is standard in nuclear physics research worldwide.
Two Neutron Sources Were Used
The team simulated two common neutron sources used in real nuclear facilities:
- Americium-241 + Beryllium (Am-Be) — a widely used lab neutron source with a broad energy spectrum.
- Californium-252 (Cf-252) — emits neutrons spontaneously. Used in medical and industrial applications.
Interestingly, the PE–TiB₂ composites blocked Cf-252 neutrons more effectively than Am-Be neutrons. This is because Cf-252 emits more fast neutrons. Fast neutrons interact more strongly with hydrogen in polyethylene. So the composite’s moderation effect is stronger for Cf-252 (Hadjal et al., 2026).
What This Means for Your Future Career
So far, this all sounds very “lab-coat and reactor” — but the applications are much broader. With this in mind, let us look at how this research (boron neutron shielding material) connects to careers students can aim for after Class 12.
Careers in and Related Fields of boron neutron shielding material
To list a few exciting paths this discovery points toward:
- Nuclear Engineer — designs shielding systems for power plants and medical reactors. Uses materials science and physics daily.
- Materials Scientist / Chemist — creates and tests new composite materials like PE–TiB₂ for safety and industrial use.
- Radiation Protection Specialist — ensures workers in nuclear and medical facilities stay safe from radiation exposure.
- Medical Physicist — applies neutron and radiation science to cancer treatment using techniques like boron neutron capture therapy (BNCT).
- Computational Physicist / Simulation Scientist — runs GEANT4 or MCNP simulations for defence, energy, and medical research.
Subjects to Focus on Right Now
Analogous to building a strong house, a strong science career needs the right foundation. To point out the most relevant subjects:
- Chemistry (Class 11–12) — atomic structure, polymers, periodic table, chemical bonding. All directly relevant to this research.
- Physics (Class 11–12) — nuclear physics chapter, radioactivity, particle interactions. Core to understanding neutron behaviour.
- Mathematics — probability and statistics underpin Monte Carlo simulations used in this very study.
- Computer Science — coding skills are essential for running simulation toolkits like GEANT4.
Balanced against all career options, nuclear engineering and materials chemistry offer some of the most impactful and well-paying roles globally. To explore how to build a career in nuclear science, visit this comprehensive article on How to Become a Nuclear Engineer at ENTECH Online.
Frequently Asked Questions (FAQs)
Titanium diboride is a hard ceramic compound made of titanium and boron. It has a very high melting point and excellent hardness. In nuclear shielding, its boron content makes it a strong absorber of thermal (slow) neutrons. You would encounter boron in your Class 11 periodic table chapter under Group 13 metalloids.
Yes, essentially. Polyethylene (PE) is one of the most common polymers on Earth. You study polymers in Class 12 organic chemistry. Its long carbon-hydrogen chains make it an outstanding neutron moderator because hydrogen nuclei are very effective at slowing down fast neutrons.
Attenuation simply means reduction in intensity. Neutron attenuation is the process of reducing the number or energy of neutrons passing through a material. A good shielding material attenuates neutrons strongly — very few make it through.
Neutron-based therapies like Boron Neutron Capture Therapy (BNCT) use boron compounds to target and destroy cancer tumours. Radiation shielding research directly supports the safety of such medical procedures. Pharmacy and pharmaceutical chemistry students interested in nuclear medicine will find this field directly connected to drug design and radiopharmaceuticals.
At this time, GEANT4 requires programming skills — typically C++. However, understanding the concept behind Monte Carlo simulations only requires basic probability and statistics, which you study in Class 11 mathematics. The actual coding is something you would learn in an undergraduate physics or engineering programme.
References (APA Style)
Hadjal, A., Saim, A., Dib, A. S. A., & Tebboune, A. (2026). Fast neutron shielding performance of polyethylene–titanium diboride (TiB₂) composites for nuclear facilities. Discover Chemistry, 3, 198. https://doi.org/10.1007/s44371-026-00659-9




