Integrated Carbon Dioxide Capture and Electrochemical Conversion
Understanding Innovation in Carbon Neutralization
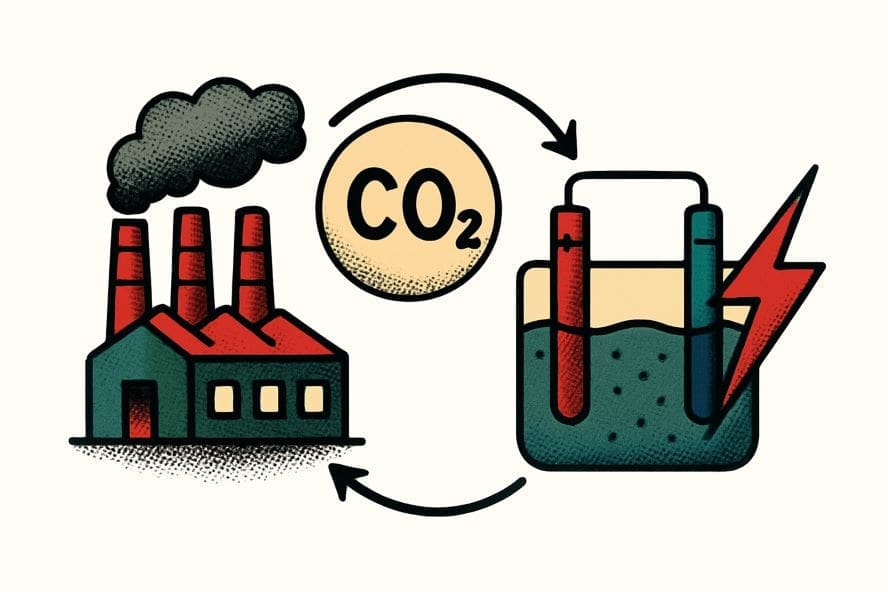
CO2 electroreduction (CO2ER) is a technology that converts atmospheric or waste Carbon dioxide capture into high value-added chemicals and fuels through renewable electricity.
Eventually, a major breakthrough within this field is the “electrochemically reactive capture” concept. It integrates CO2 absorption and conversion into a single process, effectively bypassing the massive energy penalties associated with traditional thermal regeneration.
Furthermore, this innovation extends to the creation of integrated manufacturing cycles such as retrofitting ethylene oxide (EO) plants to recycle waste Carbon dioxide capture back into ethylene, raw material used for production.
Innovator:
Key innovators identified in the sources of Carbon Dioxide Capture and Electrochemical Conversion include academic researchers such as Lei Yuan, Shaojuan Zeng, Edward H. Sargent, Magda Barecka, and Betar Gallant, alongside commercial entities like Opus 12, Siemens, Dioxide Materials, and LanzaTech.
While the innovators consists of a synergy between high-tech commercial entities and academic research labs. Leading companies include Opus 12, which has introduced 5 kW units based on established water-splitting designs. Alongside them, Siemens, Dioxide Materials, Evonik, and CERT Toronto are also involved.
Groups for example, the Innovation Academy for Green Manufacture (Chinese Academy of Sciences) and the University of British Columbia have also contributed in developing large-scale electrolyze structures and physics-based artificial intelligence models to optimize electrode performance through automated laboratories like Ada Carbon.
Practical Usage
The practical applications of this technology (Carbon Dioxide Capture and Electrochemical Conversion) are vast, as CO2ER produces chemicals that form the backbone of modern society. Ethylene oxide, a primary focus of integration studies, is an essential commodity used to produce ethylene glycol and specialty chemicals. In daily life, it serves as a critical disinfectant. Other products of this innovation include:
- Fuels: Ethanol and methane produced from CO2 capture and conversion can power aviation and marine transport, sectors where fossil fuel alternatives are currently limited.
- Chemical Feedstocks: Carbon monoxide, formate, acetate, and ethylene are using to manufacture plastics, textiles, and various industrial solvents.
Also Read: Isomers and its types
Commercial Readiness and Economic Viability
The technology is currently transitioning from laboratory research to industrial application. While it is describing as an “infant technology,” small-scale commercial projects are already operational. For instance, integrating CO2 conversion into EO manufacturing is consider economically viable in the short term, with estimated payback times of 1 to 2 years in regions with high carbon taxes or access to low-cost renewable electricity.
However, full-scale industrialization faces a stability barrier; commercial plants require systems that can operate reliably for thousands or tens of thousands of hours to ensure profitability.
As a result current research is focus on bridging the gap between lab-scale performance and the large-scale stability required for the bulk chemical market.
Future Career and Research Areas for Students
For students looking to pursue careers in this field (Carbon Dioxide Capture and Electrochemical Conversion), several critical research frontiers exist:
- Material Science and Catalysis: Researching high-performance, non-precious metal catalysts to replace expensive noble metals like silver or gold in electrodes.
- Chemical and Structural Engineering: Designing large-scale electrolyzes that address engineering hurdles like electrolyte flooding and inorganic salt crystallization which currently limit long-term operation.
- Artificial Intelligence and Physics Modeling: Developing data-driven models to navigate high-dimensional parameter spaces in automated labs, significantly accelerating the optimization of gas diffusion electrodes.
- Integrated Capture Research: Delineating the complex physicochemical properties of amine sorbents that serve simultaneously as capture media and electrolytes.
- Techno-Economic Analysis (TEA): Evaluating the profitability and life-cycle environmental impact of CCU (Carbon Capture and Utilization) technologies to guide industrial policy.
Analogy: Think of traditional Carbon Dioxide Capture and Electrochemical Conversion management as a leaky bucket where we constantly lose energy while trying to empty and refill it. This innovation is like building a recirculating fountain; instead of wasting energy to throw the water away and buy more, we use a smart pump to keep the same water flowing through the system forever, turning waste back into a beautiful and functional resource.
Additionally, to stay updated with the latest developments in STEM research, visit ENTECH Online.
Reference:
Zhu, Y., Bai, Y., Liu, J., Zuo, S., Lu, Q., Huang, C., … & Cao, G. (2023). A high-performance dual-ion battery with an ultra-high energy density of 1000 Wh kg−1. Advanced Energy Materials, 2502564. https://doi.org/10.1002/aenm.202502564