What is Hybridization and How Does It Relate to Molecular Orbitals
Hybridization and Molecular Orbit
Hybridization helps you see how atoms join together. You mix atomic orbitals to make new shapes. These new orbitals show the shape of molecules. Molecular orbitals show how electrons move in a molecule. Hybridization and molecular orbitals explain bonds in different ways. Molecular orbitals look at the whole molecule. Hybridization looks at one atom at a time.
Hybridization Basics
What Is Hybridization?
Hybridization helps explain how atoms join in molecules. Scientists like John C. Slater and Linus Pauling started this idea in 1931. They wanted to show how carbon makes four bonds in methane. Later, Robert S. Mulliken and J. H. Van Vleck made “hybridization” a common word. You mix atomic orbitals to get new hybrid orbitals. These new orbitals help you know molecule shape and bonding.
- Hybridization shows why carbon bonds look alike in methane.
- You use hybridization to guess the shape of molecules.
Formation of Hybrid Orbitals
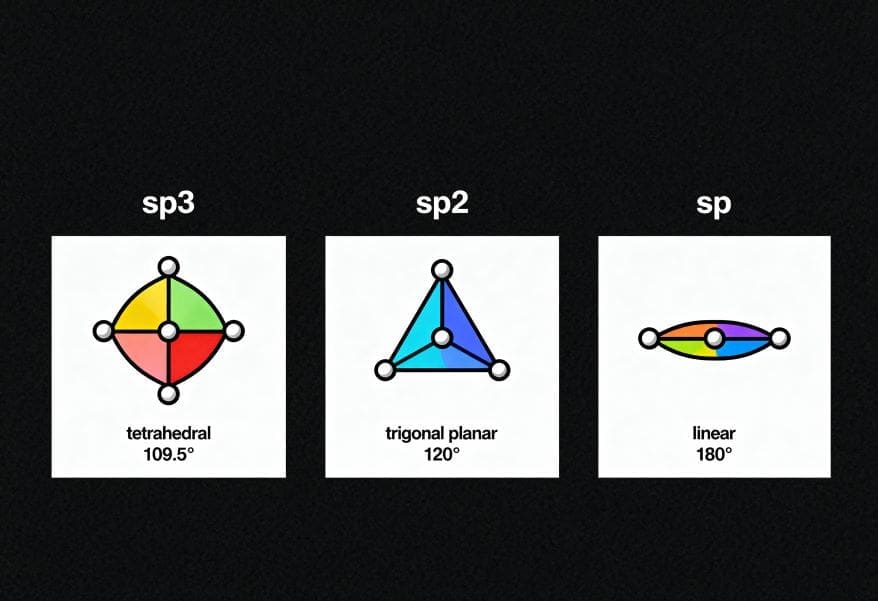
You mix atomic orbitals with close energy to make hybrid orbitals. This changes the energy levels. The new hybrid orbitals have energy between the old orbitals. For example, sp hybridization mixes one s and one p orbital. You get two sp hybrid orbitals. Each has half s-character and half p-character. You use sp3 hybridization when carbon bonds with four atoms. sp2 hybridization when carbon bonds with three atoms. sp hybridization when carbon bonds with two atoms.
Hybridization makes all hybrid orbitals have the same energy. This helps atoms bond well in molecules.
Orbital Hybridization and Molecular Geometry
Orbital hybridization helps you guess molecule shapes. In methane, carbon uses sp3 hybridization. You see four carbon-hydrogen bonds that look the same. Each bond has the same length and energy. The molecule shape is tetrahedral. In ethylene, carbon uses sp2 hybridization. The atoms make a flat shape with 120° angles.
Benzene also uses sp2 hybridization. The carbon atoms make a ring with 120° angles. You use orbital hybridization to explain these shapes.
| Molecule | Hybridization Type | Molecular Geometry |
|---|---|---|
| Methane | sp3 hybridization | Tetrahedral |
| Ethylene | sp2 hybridization | Flat, 120° angles |
| Benzene | sp2 hybridization | Flat ring, 120° angles |
Hybridization helps you guess how atoms arrange in a molecule. You see how atomic orbitals mix and make hybrid orbitals. This helps you know molecule shape and geometry.
Molecular Orbitals Overview
What Are Molecular Orbitals?
Molecular orbitals are places where electrons move in a molecule. These orbitals do not belong to just one atom. They spread out over the whole molecule. This helps you see how electrons hold atoms together.

Here is a table that shows how atomic orbitals and molecular orbitals are different:
| Feature | Atomic Orbitals | Molecular Orbitals |
|---|---|---|
| Electron Distribution | Stays with one atom | Covers the whole molecule |
| Nature of Electrons | Electrons stay in one spot | Electrons are shared by many atoms |
| Influence on Bonding | Only affects one atom | Helps keep the molecule together |
Molecular orbitals help you see how electrons change the shape and strength of molecules. This helps you know why some molecules are strong and others are weak.
Hybridization and Molecular Orbit Connection
- Orbital hybridization mixes atomic orbitals to make new hybrid orbitals.
- Hybrid orbitals help atoms join in the right shape.
- Molecular orbitals form when hybrid orbitals from different atoms overlap.
You need both orbital hybridization and molecular orbitals to explain why bonds in molecules like methane or ethylene are strong and equal.
Delocalization in Molecular Orbitals
Delocalization means electrons do not stay in one spot. In molecular orbitals, electrons can spread out over many atoms. This makes the molecule stronger. You see this in aromatic compounds like benzene. The electrons move around the ring, not just between two atoms. This sharing of electrons lowers the energy and makes the molecule tough.
Delocalization in molecular orbitals helps explain why some molecules, like benzene, are very stable.
Comparing Hybridization and Molecular Orbit Theory
Localized vs. Delocalized Bonding
You can use hybridization or molecular orbit theory to show how atoms bond. Hybridization theory mixes atomic orbitals on one atom. This makes new orbitals that help atoms bond. These bonds are called localized bonds. Localized bonds happen between two atoms, like carbon and hydrogen in methane.
Molecular orbital theory uses delocalized bonding. Electrons move all over the molecule. They do not stay between just two atoms. Delocalized electrons can make molecules stronger. You see this in benzene and ozone. Electrons spread out and touch many atomic nuclei.
- Delocalized bonding lets electrons touch more than one nucleus. This makes molecules stronger.
- Localized models, like valence bond theory, might miss some strength. They do not show electron delocalization.
- Molecular orbital theory explains color, resonance, and strength of molecules. Localized models cannot do this.
You can find proof of delocalized electrons in ozone. The table below shows how scientists know electrons spread out in ozone.
| Evidence Description | Details |
|---|---|
| Molecular Orbitals | p orbitals overlap in ozone and show electron delocalization. |
| Bonding and Nonbonding MOs | Electron density spreads out in the molecule. |
| Spectral Absorption | Ozone takes in UV light because of delocalized electrons. |
Predicting Shapes and Properties
Hybridization and molecular orbit theory help you guess shapes and properties of molecules. Hybridization theory uses atomic orbitals to explain bond angles. You see this in water and carbon dioxide. Valence bond theory says water should have a 90° angle. Experiments show the angle is 104.5°. Hybridization theory uses four hybrid orbitals on oxygen. These point to the corners of a tetrahedron. The predicted angle is 109.5°, which is close to the real angle.
| Evidence Description | Details |
|---|---|
| Bond Angle in Water | Real angle is 104.5°, not 90°. Valence bond theory does not match. |
| Hybridization in Water | Four hybrid orbitals on oxygen predict a 109.5° angle. |
Strengths and Limitations
Hybridization theory works well for simple molecules. You can use it for methane, water, and ammonia. It helps you see how carbon and hydrogen atoms bond. You can guess shapes and bond angles.
Molecular orbital theory works better for harder molecules. You use it for conjugated systems, like benzene. It explains delocalized electrons and resonance. You see how p orbitals overlap and let electrons move across the molecule. The Hückel approximation helps you study pi molecular orbitals. You can split sigma and pi bonding frameworks.
Practical Uses and Limitations
When to Use Hybridization
You use hybridization to guess a molecule’s shape. This theory shows how atomic orbitals mix on one atom. You can match the shape to the type of hybridization. For example, a straight line shape uses sp hybridization. A triangle shape uses sp² hybridization. A four-corner shape uses sp³ hybridization. The table below helps you match shapes and hybridization:
| Molecular geometry | Hybridization |
|---|---|
| linear | sp |
| trigonal | sp² |
| tetrahedral | sp³ |
| trigonal bipyramidal | sp³d |
| octahedral | sp³d² |
When to Use Molecular Orbitals
You use molecular orbital theory to study how strong bonds are. This theory shows how electrons move in the whole molecule. You use it to learn about magnetism and resonance.
The molecular orbital idea for O₂ shows why liquid oxygen is pulled into a magnetic field. Two unpaired electrons make it paramagnetic.
Molecular orbital theory to see how atomic orbitals join and make molecular orbitals. You use this theory for molecules with delocalized electrons, like benzene. You also use it for conjugated systems and to guess color and strength.
Real-World Applications
You use hybridization and molecular orbital theory in many fields.
Chemists use these ideas to make new medicines. Molecular orbital theory helps them change drug designs and make drugs work better. It also helps guess how drugs act in the body. Hybridization helps chemists know the shape of molecules like carbon-based drugs.
Molecular orbital theory helps scientists study solids. The COOP analysis lets them see how atoms join in materials. Linus Pauling used both theories to explain benzene’s structure. His work helped chemists learn about electronegativity and molecular structure. Scientists used these ideas to study DNA. Hybridization and molecular orbital theory help you understand bonds in carbon compounds, hydrogen bonds, and many other molecules.
You can use hybridization and molecular orbital theories to learn about chemical bonds. Hybridization helps you guess the shape and bond angles in carbon compounds. Molecular orbital theory shows how electrons move in the whole molecule. Both ideas help explain why carbon bonds are strong and why molecules like benzene do not break apart easily.
Chemists use both hybridization and molecular orbital theory all the time. These ideas are important in making new medicines, changing enzymes, and building new materials.
Key Takeaways
- Hybridization shows how atoms join by mixing atomic orbitals. This makes new shapes and helps us guess how molecules look.
- Molecular orbitals show how electrons move in the whole molecule. They help us understand how strong and stable bonds are.
- Use hybridization to find the shape of simple molecules like methane. Molecular orbital theory works better for harder molecules like benzene.
- When electrons spread out in molecular orbitals, some molecules get stronger. This makes those molecules more stable.
- Hybridization and molecular orbital theory are both important in chemistry. They help us learn about bonding, guess properties, and make new materials.
FAQ
What does hybridization mean in chemistry?
Hybridization is when atomic orbitals mix together. This makes new orbitals. These new orbitals help atoms bond. You can use them to guess molecule shapes.
Why do you need hybridization to predict molecular shapes?
Hybridization shows how atoms fit in a molecule. You match the hybridization type to the shape. For example, sp3 hybridization makes a tetrahedral shape.
How does hybridization differ from molecular orbital theory?
Hybridization looks at one atom at a time. You mix orbitals on that atom only. Molecular orbital theory looks at the whole molecule. Electrons move over many atoms.
Can you use hybridization for all molecules?
Hybridization works for simple molecules like methane and water. For bigger molecules with delocalized electrons, use molecular orbital theory. Hybridization does not show electrons moving across many atoms.
What are some signs that hybridization is happening?
You see equal bond lengths and angles in molecules. Methane has four equal bonds. This shows sp3 hybridization. You also see shapes like tetrahedral or trigonal planar.
Additionally, to stay updated with the latest developments in STEM research, visit ENTECH Online. Basically, this is our digital magazine for science, technology, engineering, and mathematics. Also, at ENTECH Online, you’ll find a wealth of information.
Reference
Flowers, P., Theopold, K., Langley, R., & Robinson, W. R., PhD. (2019, February 14). 8.2 Hybrid Atomic Orbitals – Chemistry 2E | OpenStax. https://openstax.org/books/chemistry-2e/pages/8-2-hybrid-atomic-orbitals




