Your Morning Coffee Could Power the Next Generation of Batteries
Estimated reading time: 6 minutes
The Hook: Waste in Your Cup, Power in Your Phone
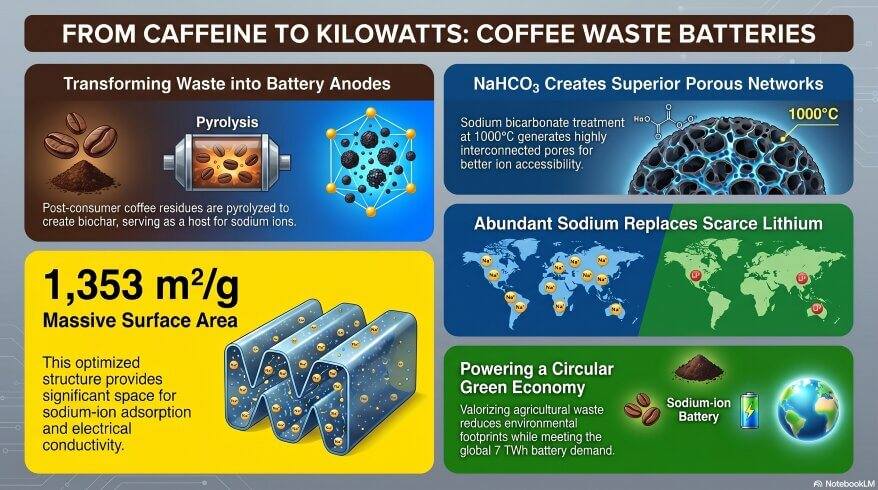
Think about the last time someone made coffee at home. After brewing, the used coffee grounds go straight into the bin. Most people never think twice about it. But a group of scientists from Mexico did — and what they found could change how we build batteries forever. This sounds too good to be true. After all, how can soggy coffee waste become a key material in next-generation energy technology? All things considered, the answer lies in chemistry. Seeing that you already know about carbon structures, chemical reactions, and thermodynamics, this research by Escobar Juárez, O.A. and team will feel surprisingly familiar.
Key Takeaways: Waste to Energy
- Scientists turned used coffee grounds into battery materials called biochar.
- These biochars work as anodes in sodium-ion batteries (SIBs).
- The best result came from treating coffee waste with NaHCO₃ at 1000 °C.
- This biochar had a surface area of 1353 m²/g — enormous at the nanoscale.
- SIBs are a greener, cheaper alternative to lithium-ion batteries.
- This research links directly to your Grade 11 and 12 chemistry concepts.
What Did the Researchers Actually Do?
Escobar Juárez et al. (2026) published a study in Chemistry (MDPI). They collected post-consumer coffee grounds — the kind thrown away after making a cup. They dried them. Then they soaked the grounds in two common salt solutions: sodium chloride (NaCl) and sodium bicarbonate (NaHCO₃). After that, they heated the samples in a furnace without oxygen. This process is called pyrolysis. They tested two temperatures: 500 °C and 1000 °C.
What Is Pyrolysis?
Pyrolysis is a thermochemical process. To explain it simply:
- Organic matter heats up in zero-oxygen conditions.
- Carbon-rich solid (called biochar) forms.
- Gases and liquids also release during the process.
- The carbon structure of the biochar depends on temperature.
Your Grade 12 chemistry covers chemical bonding and carbon structures. So this is not new territory for you. While it may be true that pyrolysis sounds industrial, the chemistry behind it is the same carbon science you study in class.
Why Sodium Bicarbonate Worked Best: Waste to Energy
The researchers tested both NaCl and NaHCO₃. The NaHCO₃ sample heated to 1000 °C gave the best result. To put it differently, here is what happened chemically:
- At ~500 °C: NaHCO₃ → Na₂CO₃ + H₂O + CO₂ (large but sparse pores formed)
- At ~1000 °C: Na₂CO₃ → Na₂O + CO₂ (CO₂ etched tiny micropores into the carbon)
- Na₂O also reacted with the carbon surface, opening interconnected channels.
As a result, the final material had a surface area of 1353 m²/g. That is larger than a football field — packed into just one gram of material. What’s more, the pore size was 2.6 nm. This is exactly right for sodium ions (Na⁺) to pass through.
Why Do We Even Need Sodium-Ion Batteries? Waste to Energy
At the present time, most phones, laptops, and electric vehicles use lithium-ion batteries. Lithium works well. But it has problems. To enumerate the key issues:
- Lithium is expensive and difficult to mine.
- Its geographic distribution is unequal. Only a few countries hold most reserves.
- Mining lithium causes serious environmental damage.
- Demand is growing faster than supply, especially with rising EV adoption.
Balanced against these problems, sodium offers a powerful alternative:
- Sodium is one of the most abundant elements in Earth’s crust.
- It is found in seawater, salt, and many minerals.
- It is far cheaper to extract and process.
- It is more evenly distributed around the world.
What Were the Results?
The scientists tested the biochar samples as battery anodes (the negative electrode). They built small electrochemical cells and ran charge-discharge cycles. Here is a summary of the key findings:
NaHCO₃-Treated Biochar at 1000 °C (Best Performer)
- Surface area: 1353.22 m²/g
- Pore volume: 0.83 cm³/g
- Pore size: 2.6 nm (mesopore range — perfect for Na⁺)
- Initial capacity: 34 mAh/g
- Stability: Remained stable for more than 15 charge-discharge cycles
- Structure: Mostly amorphous carbon with graphitic domains
NaCl-Treated Biochar at 500 °C (Weaker Performer)
- Initial capacity was 70 mAh/g at first.
- But it dropped sharply after the very first cycle.
- It stabilised at only 10 mAh/g.
- The pore structure was irregular and poorly connected.
- Na⁺ could not move through the material efficiently.
So as to summarise the comparison: NaHCO₃ biochar was more stable. NaCl biochar had a higher initial number but fell apart fast. In electrochemistry, stability over many cycles is far more important than a one-time high number. This is called cycling stability, and it is what makes a battery commercially useful.
What Does This Mean for You — And for Your Future Career?
Above all, this research shows something exciting. Everyday waste to Energy materials can become cutting-edge technology components.You need clever chemistry.
Provided that you are thinking about studying further after Grade 12, this research opens a window into several amazing career paths. Here are some of them:
Career Paths This Research Connects To
- Pharmaceutical and Industrial Chemistry — understanding how carbon-based materials interact with ions is also used in drug delivery systems where porous carbon carries medicine inside the body.
- Materials Science and Nanotechnology — designing materials at the nanoscale (like 2.6 nm pores) is one of the fastest-growing fields in science.
- Green Chemistry and Sustainability — turning waste into useful products is a growing area. The pharmaceutical and chemical industry need sustainable processes.
- Electrochemistry and Battery Research — electric vehicles, grid storage, and portable electronics all need better batteries. Chemists are at the centre of this.
- Chemical Engineering — scaling up lab results into real products is what chemical engineers do. This research is a perfect example of lab-to-industry science.
If you are interested in learning more about energy storage innovations, check out this article on Flow Batteries: What You Need to Know for a broader look at next-gen energy storage. You can also read about Biodegradable Batteries: A Green Future for Wearable Tech to see how sustainable battery chemistry applies to wearables and medicine.
Frequently Asked Questions on Waste To Energy
Biochar is a carbon-rich solid. It forms when organic material (like wood, plant waste, or coffee grounds) heats up in zero-oxygen conditions. The process is called pyrolysis.
A sodium-ion battery stores and releases energy using the movement of sodium ions (Na⁺) between the anode and cathode. It works similarly to a lithium-ion battery. But it uses sodium instead.
Surface area means how much reactive material is available at the nano level. A higher surface area means more places for sodium ions to interact and store. 1353 m²/g in one gram of material is enormous at this scale.
Sodium is far more abundant. It is cheaper to mine. It is found all over the world, not just in a few countries. This makes it a more sustainable and affordable option for large-scale energy storage.
Pyrolysis is a thermochemical decomposition. It breaks down organic compounds using high heat but no oxygen. It produces solid biochar, gases, and liquid by-products.
Reference
Escobar Juárez, O. A., Del Angel Meraz, E., Quiroga González, E., Osorio García, M., Pacheco Sosa, J. G., Pantoja Castro, M. A., & Hernández Cruz, M. G. (2026). Biochar synthesized from post-consumer coffee waste using molten salts for sodium-ion battery applications. Chemistry, 8(4), 51. https://doi.org/10.3390/chemistry8040051




