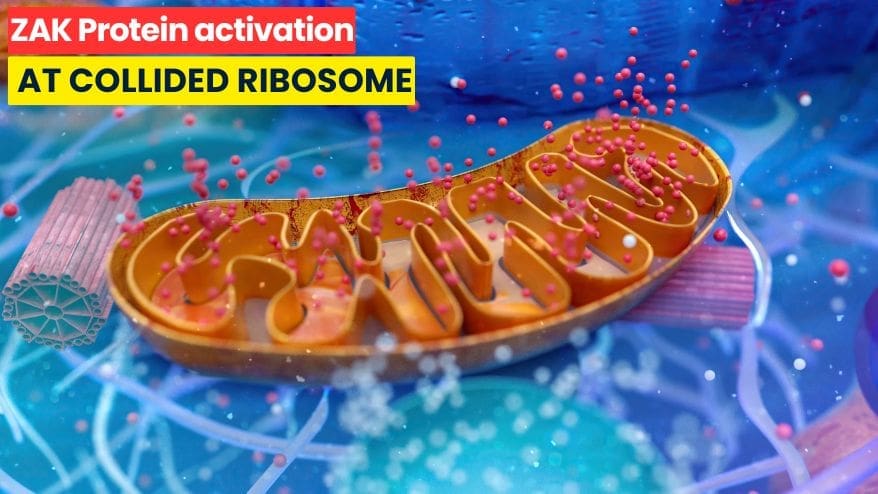
How ZAK Protein Detects Ribosome Collisions to Trigger Cell Stress Responses
Cells rely on ribosomes to translate mRNA into proteins. Often, multiple ribosomes work on the same mRNA strand, forming polysomes. However, when ribosomes face problems like nutrient deprivation or chemical damage, they stall. ZAK Protein Detects Ribosome Collisions from these stalls, where a trailing ribosome collides with the stalled one. These collisions send important signals that trigger cellular quality control and stress responses.
ZAK Protein Detects Ribosome Collisions and Triggers Cellular Signaling
The ribosome is critical for translating mRNA into protein. Often, multiple ribosomes work together on a single mRNA, forming structures called polysomes. When cells face stress such as nutrient shortage or chemical damage, ribosomes can stall during translation. Importantly, stalled ribosomes can collide with trailing ones. These ribosome collisions send important signals inside the cell.
One key player in this process is the protein kinase called ZAKα (simply ZAK). ZAK acts as a sensor that detects these collisions and triggers the cellular response. This is known as the ribotoxic stress response (RSR). It activates downstream signaling pathways such as SAPKs p38 and JNK, which can halt the cell cycle or promote cell death if necessary. On this basis alone, understanding how ZAK recognizes stalled and collided ribosomes matters greatly for biology.
ZAK Interacts with Ribosomes via RACK1 Interface
Recent research using cryo-electron microscopy has provided clear insights into how ZAK binds collided ribosomes at their interfaces. Scientists found that ZAK targets a scaffold protein named RACK1, which resides on the 40S small ribosomal subunit exactly where two stalled ribosomes collide.
ZAK contains several domains such as an N-terminal kinase domain and SAM domains that form dimers bridging two RACK1 proteins from collided ribosomes. This interaction connects stalled and trailing ribosomes through ZAK’s structured regions including a RACK1-interacting motif (RIM). As a result, it stabilizes ZAK’s position at these collision sites preparing it for activation.
The C-terminal tail of ZAK Anchors to 40S Subunit
The analysis also revealed another important finding: the C-terminal domain (CTD) of ZAK has positively charged residues crucial for anchoring to the 40S subunit near proteins eS27 and eS26 within RACK1. This region acts somewhat like a pin holding onto specific parts of each individual ribosome regardless of whether collisions occur or not, positioning ZAK nearby to sense translation problems quickly.
“The structural details show how molecular recognition adapts to stress conditions,” noted Dr. Smith, one of the lead researchers in this study.
ZAK Activation Leads to Vital Cellular Outcomes
Once bound at collision sites, ZAK undergoes autophosphorylation activating its kinase function. This activation launches signaling cascades which help cells arrest their growth or undergo apoptosis depending on damage severity. Control experiments indicated that inactive mutants of ZAK remained stuck on polysomes but could not trigger these responses.
This balance is important because persistent translation errors cause cellular dysfunction while efficient quality control systems protect genome integrity and proteostasis – all mediated partly by the effective detection by proteins like ZAK upon collision events.
A Path Forward in Ribosome Collision Research
The findings described here represent significant progress elucidating how cells monitor translation fidelity under stress through direct molecular interactions involving scaffold proteins such as RACK1 combined with kinases like ZAK. Such knowledge opens doors for future research aimed at targeting diseases linked to protein misfolding or defective stress responses including cancer or neurodegeneration.
Additionally, to stay updated with the latest developments in STEM research, visit ENTECH Online. Basically, this is our digital magazine for science, technology, engineering, and mathematics. Also, at ENTECH Online, you’ll find a wealth of information.
Reference
- Huso, V. L., Saba, J. A., Becker, T., & Green, R. (2025). ZAK activation at the collided ribosome. Nature. https://doi.org/10.1038/s41586-025-09772-8